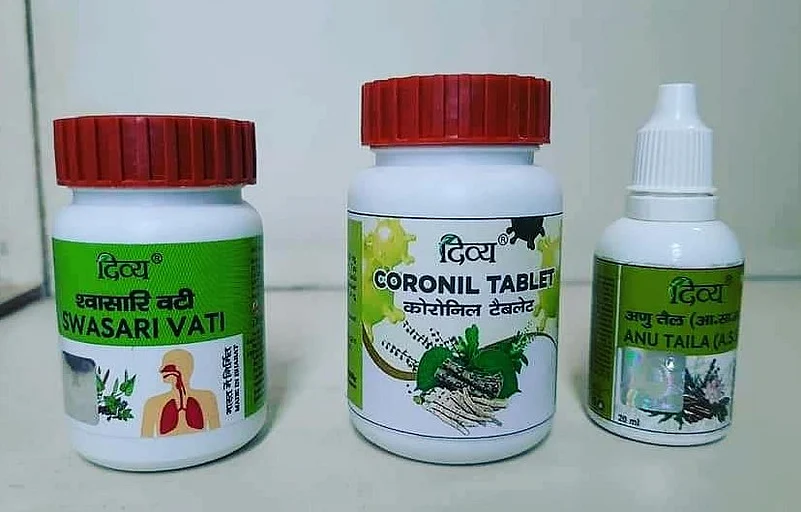
The Union Ministry of Ayush on Tuesday sought details of an Ayurvedic drug, touted as Covid-19 cure, hours after it was launched by Yoga guru Ramdev's Patanjali Ayurved Ltd. The Ministry has asked the firm to stop advertisement till the drug and claims about it are examined.
The firm claimed that their two Ayurved-based medicines have shown 100 per cent favourable results during clinical trials on COVID-19 infected patients except those on a life support system.
"Facts of the claim and details of the stated scientific study are not known to the Ministry," the statement released by the Union ministry said.
"The concerned Ayurvedic drug manufacturing company has been informed that such advertisements of drugs including Ayurvedic medicines are regulated under the provisions of Drugs and Magic Remedies (Objectionable Advertisements) Act, 1954 and Rules thereunder and the directives issued by the Central Government in the wake of COVID outbreak," it further said,
To verify the claims made by Patanjali Ayurved, the Ayush Minishtry asked the firm to provide at the earliest "details of the name and composition of the medicines being claimed for COVID treatment; site(s)/hospital(s), where the research study was conducted for COVID-19; protocol, sample size, Institutional Ethics Committee clearance, CTRI registration and results data of the study (ies) and stop advertising/publicizing such claims till the issue is duly examined."
Moreover, the Ministry has requested concerned State Licensing Authority of Uttarakhand government to provide copies of license and product approval details of the Ayurvedic medicines being claimed as treatment of COVID -19.
The medicines have been developed by Patanjali Research Center, Haridwar and privately-owned National Institute of Medical Science, Jaipur following all protocols with clinically controlled trial-based evidence, Ramdev told PTI.
"Patanjali first conducted clinical case study and conducted clinical control trials following all protocols of drug discovery," he said.
A government notification bars companies from advertising a cure without government approval.
Sidestepping questions on the drug being approved by government agencies such as ICMR, Ramdev said clinical controlled study of these medicines was done in several cities including Delhi, Ahmedabad and Meerut and the RCT (Randomized Clinical Trial) controlled with placebo was conducted at Jaipur-based National Institute of Medical Sciences & Research.
"This was done after getting approval from Clinical Trial Registry of India (CTRI) and all other required formalities," he said. "We have followed all the parameters set up by modern science for such clinical trials."
(With PTI inputs)