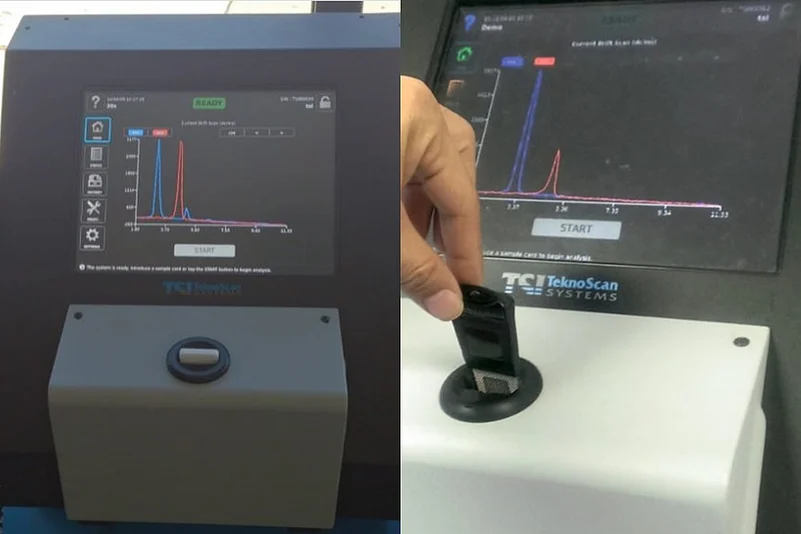
Your breath can diagnose your Covid-19 status within 20 seconds. This innovative technology can be a game changer for India in its fight against the pandemic.
However, its inventor has been running from pillar to post to get approval for public use for past more than a month but there is no government body competent to validate the technology.
India’s premier research organisation and a nodal body for Covid-19 management Indian Council for Medical Research (ICMR) say that they don’t have the expertise to assess the technology.
A Gurugram based Anika Sterilis Pvt. Ltd which works in collaboration with Canada-based Teknoscan Systems Inc has developed this machine and its trial video is viral on WhatsApp.
Here, a person needs to breathe out five to six times on a coated sterilized small-sized sample card and the card is immediately inserted into a machine. It takes 20 seconds to tell whether the person is Covid-19 positive or negative.
The technology is based on the principle of identifying disease-specific Volatile Organic Compound (VOC).
Infected persons breathe out certain chemical compounds that are unique to Covid-19 disease. These are also called biomarkers.
“There are several volatile organic compounds (VOCs) mainly consisting of aldehydes, ketones, and esters that have been identified and reported in several studies in China and the UK,” Hemant Kumar, Managing Director, Anika Sterilise Pvt. Ltd said.
He added, “The identification has been made using gas chromatography and mass spectrometry with extensive library search to confirm the identity of the key compounds associated with people infected with the coronavirus.”
Claiming to develop the world's first Covid-19 fully automated breath analyzer, he says that the machine has been programmed to take samples of a patient's breath to detect COVID-19's specific volatile organic compounds (VOCs).
Hopeful to get immediate approval from the government, he first applied with the ICMR. He was surprised to get a reply that they didn’t have the expertise to evaluate such a technology.
In the reply, Dr Madhumathi from the ICMR provided him a list of laboratories with their field of competence under the Department of Biotechnology and the Council for Scientific and Industrial Research (CSIR).
None of the labs have been shown in the reply to have the expertise to deal with VOCs technology.
“I am not asking the government to accept my machines. I am telling them to at least take a look at the technology and then take a call. But they are not even ready to do that,” Kumar said.
As per the present legal provision, Kumar can use his machine for trial purposes by merely intimating it to the Central Drugs Standard Control Organisation (CDSCO) in writing but he can’t use it unless ICMR gives its nod.
“My machine is already on trial at two places in Delhi and we have tested over 700 people now. The result has been extremely encouraging. Many top companies are impressed with the performance but official approval is the biggest hindrance,” he said.
He added, “I want to help the country fight against the disease but it looks like I have to fight the system first and win over it before fighting against Sars_Cov_2,” he said.
Kumar also said that he is working to retrofit mobile vans with the detector unit and provide onsite testing for coronavirus-infected people.
“It is a quick first screening approach to identify infected persons with the virus for further follow-up, validation, and treatment,” he said.